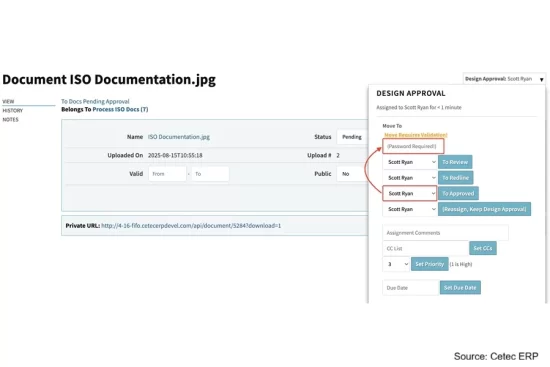
Cetec ERP released versions 4.19 and 4.20 designed with new capabilities to support the day-to-day compliance and quality needs of medical device and life science companies.
Related Posts
VIDEO PODCAST | How to Increase Speed Without Compromising Quality in Aerospace Manufacturing
Trevor Campbell, Sales Development Manager of Microscopy at Zeiss Industrial Quality Solutions, discusses artificial intelligence, microscopy, and a…
System Integration Testing (SIT) – A Detailed Overview
Content 1. An Overview of System Integration Testing 2. Importance of System Integration Testing 3. Benefits of System…
PODCAST | Standards: Waiting With Breathless Anticipation
Andy Nichols is a managing partner of Quality Nation Incorporated with more than 40 years of experience in…



