In 2025, medical device manufacturers are grappling with heightened regulatory uncertainty, leading to slower reviews and communication difficulties with the FDA. This situation drives them to reassess operations for compliance and risk management, with a strong emphasis on maintaining product quality, which is vital for patient safety.
Why it Pays to Prioritize Quality Amid Regulatory Uncertainty

Related Posts
Embracing Innovation: Adapting to Evolving Control Systems Trends
The relentless pursuit of quality and reliability is at the core of all manufacturing operations. As control systems…
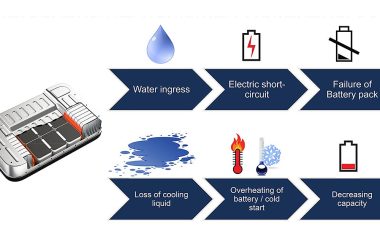
Best Practice Nondestructive Testing for EV Battery Packs, the Equivalent Channel Method
In 2023, SAE International’s Battery Standards Testing Committee, consisting of over 20 companies in EV battery manufacturing and…
Medical Device Q&A
Sai Ranjith Ramakrishnan Kumar (Sai) is a medical device quality engineering expert, Six Sigma Green Belt Certified, and…